What are you looking for?
Search
Tirzepatid 30mg*10vials
230.00
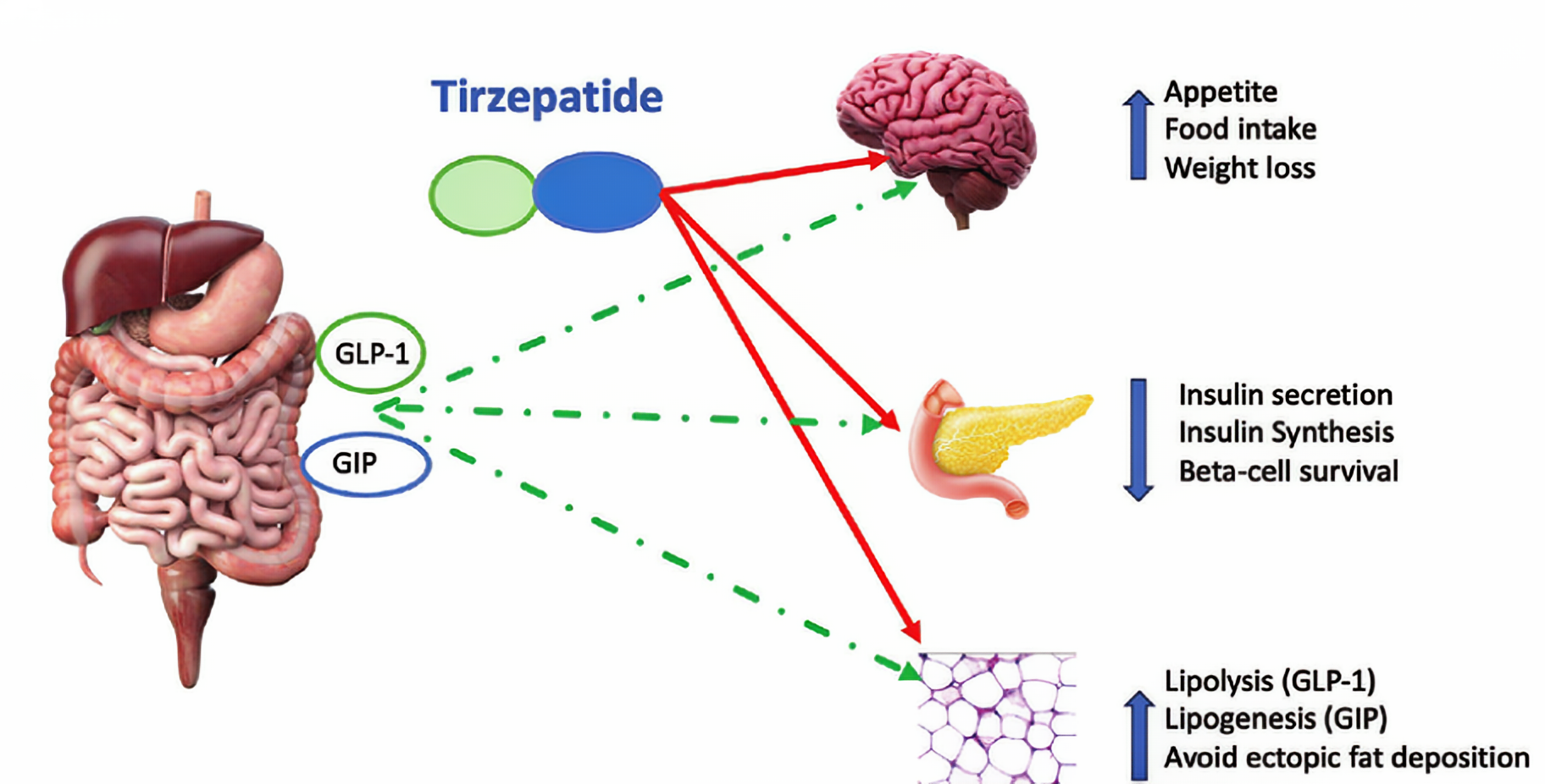
Tirzepatid is a novel dual-target agonist that primarily acts on the glucagon-like peptide-1 receptor (GLP-1R) and the glucose-dependent insulinotropic polypeptide receptor (GIPR). It mimics the action of naturally occurring intestinal insulin-secreting hormones in the body, increasing insulin secretion, inhibiting glucagon secretion, and slowing gastric emptying, thereby effectively controlling blood glucose levels. Additionally, Tirzepatid regulates the central nervous system, increasing feelings of fullness and reducing food intake, leading to significant weight loss. It also improves lipid metabolism to some extent and reduces the risk of cardiovascular disease. Tirzepatid is primarily used to treat type 2 diabetes and obesity. For patients with type 2 diabetes, it not only effectively lowers blood sugar but also reduces the risk of cardiovascular complications. In obesity treatment, it helps patients lose weight and improve obesity-related metabolic disorders. Tirzepatid also has therapeutic effects in metabolic diseases such as non-alcoholic fatty liver disease.
The peptide will be provided as lyophilized powder to ensure maximum stability.