What are you looking for?
Search
What is Mazdutide?
ALL ARTICLES AND PRODUCT INFORMATION PROVIDED ON THIS WEBSITE ARE SOLELY FOR INFORMATION DISSEMINATION AND EDUCATIONAL PURPOSES.
The products provided on this website are intended exclusively for in vitro research. In vitro research (Latin: *in glass*, meaning in glassware) is conducted outside the human body. These products are not pharmaceuticals, have not been approved by the U.S. Food and Drug Administration (FDA), and must not be used to prevent, treat, or cure any medical condition, disease, or ailment. It is strictly prohibited by law to introduce these products into the human or animal body in any form.
As the incidence of metabolic diseases such as overweight, obesity, and diabetes continues to rise globally, the search for more effective therapeutic drugs has become a hot topic in the medical field. Mazdutide, as a novel dual receptor agonist, can simultaneously activate the GLP-1 receptor and the glucagon receptor, and is expected to play a unique role in weight loss and blood sugar control, offering new opportunities for the treatment of metabolic diseases.

Figure 1 The chemical structure of Mazdutide.
Overview of Mazdutide
Mazdutide is a GLP-1/glucagon dual receptor agonist, a novel drug for the treatment of metabolic diseases. Over the past few decades, drug development targeting metabolic diseases has continued to advance. GLP-1 receptor agonists and glucagon receptor agonists have demonstrated certain therapeutic effects when used alone, but they also have their own limitations. To seek a more ideal treatment option, researchers have focused on developing drugs that can act on multiple targets simultaneously, leading to the creation of Mazdutide. Its development process has undergone rigorous preclinical studies and multi-stage clinical trials, gradually validating its safety and efficacy.
Mechanism of Action
(1) Activation of the GLP-1 Receptor
Regulation of blood glucose homeostasis: GLP-1 is a peptide hormone secreted by intestinal L cells. After activating the GLP-1 receptor, Mazdutide regulates blood glucose through multiple pathways. It promotes insulin secretion by pancreatic β cells, enhances insulin sensitivity, thereby accelerating glucose uptake and utilization, and lowering blood glucose levels. It inhibits glucagon secretion from pancreatic α cells and reduces hepatic glucose output, further maintaining blood glucose stability.
Delaying Gastric Emptying: Mazdutide activates the GLP-1 receptor, acting on the gastrointestinal nervous system to slow gastric emptying. This prolongs food retention in the stomach, increasing satiety and reducing food intake, thereby aiding weight control.
Regulation of appetite: After GLP-1 receptor activation, signals are transmitted to the central nervous system, acting on the hypothalamic feeding center to regulate the secretion of appetite-related neuropeptides, such as reducing neuropeptide Y (NPY) expression and increasing proopiomelanocortin (POMC) expression, thereby inducing a sense of fullness and reducing food intake.
(2) Activation of glucagon receptors
Promoting energy expenditure: When the glucagon receptor is activated by Mazdutide, it promotes fat breakdown and fatty acid oxidation, increasing energy expenditure. Additionally, it upregulates the expression of uncoupling protein 1 (UCP1), promoting thermogenesis in brown adipose tissue, further enhancing the body's energy metabolism and aiding in weight loss.
Regulation of glucose metabolism: Activation of glucagon receptors promotes hepatic glycogenolysis and gluconeogenesis. However, under the influence of Mazdutide, this promotional effect does not simply elevate blood glucose levels but instead achieves precise blood glucose regulation through synergistic interaction with GLP-1 receptor activation effects. When blood glucose levels are high, the hypoglycemic effect mediated by GLP-1 receptor activation predominates; when blood glucose levels are low, the hyperglycemic effect mediated by glucagon receptor activation prevents hypoglycemia.
(3) Synergistic Mechanism
Mazdutide simultaneously activates both GLP-1 receptors and glucagon receptors. The signaling pathways activated by these two receptors work synergistically to exert a more comprehensive and powerful metabolic regulatory effect. In terms of weight loss, GLP-1 receptor activation reduces energy intake by suppressing appetite and delaying gastric emptying, while glucagon receptor activation promotes energy expenditure. The synergistic action of both receptors more effectively achieves weight reduction. In terms of blood glucose regulation, the effects of both receptor activations complement each other to maintain blood glucose within the normal range and avoid excessive fluctuations.
3. Physiological Effects
(1) Weight Loss Effects
Clinical evidence: Multiple clinical trials have demonstrated that Mazdutide has significant weight loss effects. In studies targeting overweight or obese adults in , 24 weeks of Mazdutide treatment (with a maximum dose of 6 mg) resulted in an average weight loss of 6.7%–11.3%, while the placebo group experienced a 1.0% weight gain. Among different dose groups, Mazdutide at 4.5 mg and 6 mg demonstrated more pronounced weight loss effects, with treatment differences compared to placebo ranging from -7.7% to -12.3% (P < 0.0001). In a systematic review and meta-analysis involving 680 participants, Mazdutide demonstrated a significant advantage over placebo in weight reduction, with a mean difference (MD) of -6.22% (95% confidence interval [CI]: -8.02% to -4.41%).

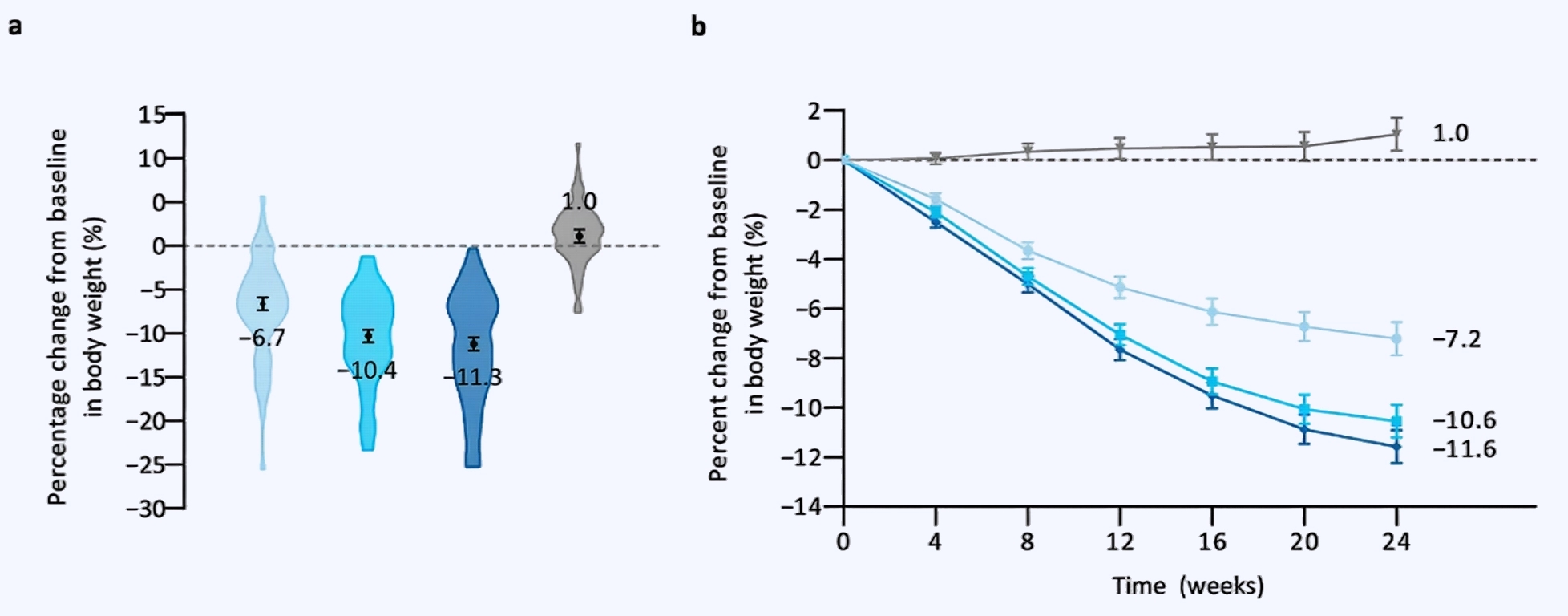
Figure 2 Body weight efficacy endpoints. a Percentage change from baseline in body weight at week 24. b Percentage change from baseline in body weight over time.
(2) Glycemic Regulation Effects
Effects on patients with type 2 diabetes: For patients with type 2 diabetes, Mazdutide effectively reduces hemoglobin A1c (HbA1c) and fasting blood glucose levels. In a related randomized, double-blind, placebo-controlled Phase 2 clinical trial, patients with type 2 diabetes who received Mazdutide treatment (at a maximum dose of 6 mg) for 20 weeks, showed an average change in HbA1c ranging from -1.41% to -1.67%, while the placebo group showed a change of 0.03%, with a statistically significant difference compared to the placebo (P < 0.0001). Patients also experienced a significant decrease in body weight, with the average percentage change showing a dose-dependent relationship, reaching up to -7.1%.
Advantages in blood glucose regulation: Mazdutide not only lowers blood glucose levels but also improves blood glucose variability and reduces the risk of hypoglycemic events. Compared with traditional antidiabetic drugs, its dual receptor agonist mechanism of action achieves more precise and comprehensive blood glucose regulation, thereby improving patients' quality of life and long-term prognosis.
(3) Effects on cardiovascular and metabolic indicators
Blood pressure regulation: Mazdutide reduces both systolic and diastolic blood pressure. Systematic reviews and meta-analyses show that compared with placebo, Mazdutide reduces systolic blood pressure by an average difference (MD) of -7.57 mmHg (95% CI: -11.17 to -3.98 mmHg); for diastolic blood pressure reduction, the MD was -2.98 mmHg (95% CI: -5.74 to -0.22 mmHg). This may be attributed to Mazdutide's multiple mechanisms, including improving vascular endothelial function, reducing peripheral vascular resistance, and regulating water-sodium balance.
Lipid regulation: Mazdutide effectively regulates lipid profiles. It reduces total cholesterol (MD = -16.82%, 95% CI: -24.52 to -9.13%), triglycerides (MD = -43.29%, 95% CI: -61.57 to -25.01%), and low-density lipoprotein (MD = -17.07%, 95% CI: -25.54 to -8.60%) levels, while also exerting a certain regulatory effect on high-density lipoprotein (MD = -7.54%, 95% CI: -11.26 to -3.83%). These effects help reduce the risk of cardiovascular disease and have a protective effect on the cardiovascular system.
(4) Effects on hyperuricemia
Animal experimental evidence: In a hyperuricemia (HUA) rat model study, subcutaneous injection of 0.05 mg/kg and 0.075 mg/kg of Mazdutide (every 3 days) significantly reduced serum uric acid (SUA) levels in rats. Compared with the HUA group, SUA and serum creatinine (SCr) levels were significantly reduced in the Maz-MD and Maz-HD groups, and urinary protein (U-Pro) levels were also significantly reduced in the Maz-MD group. Additionally, Mazdutide improved renal histopathological changes in HUA rats.

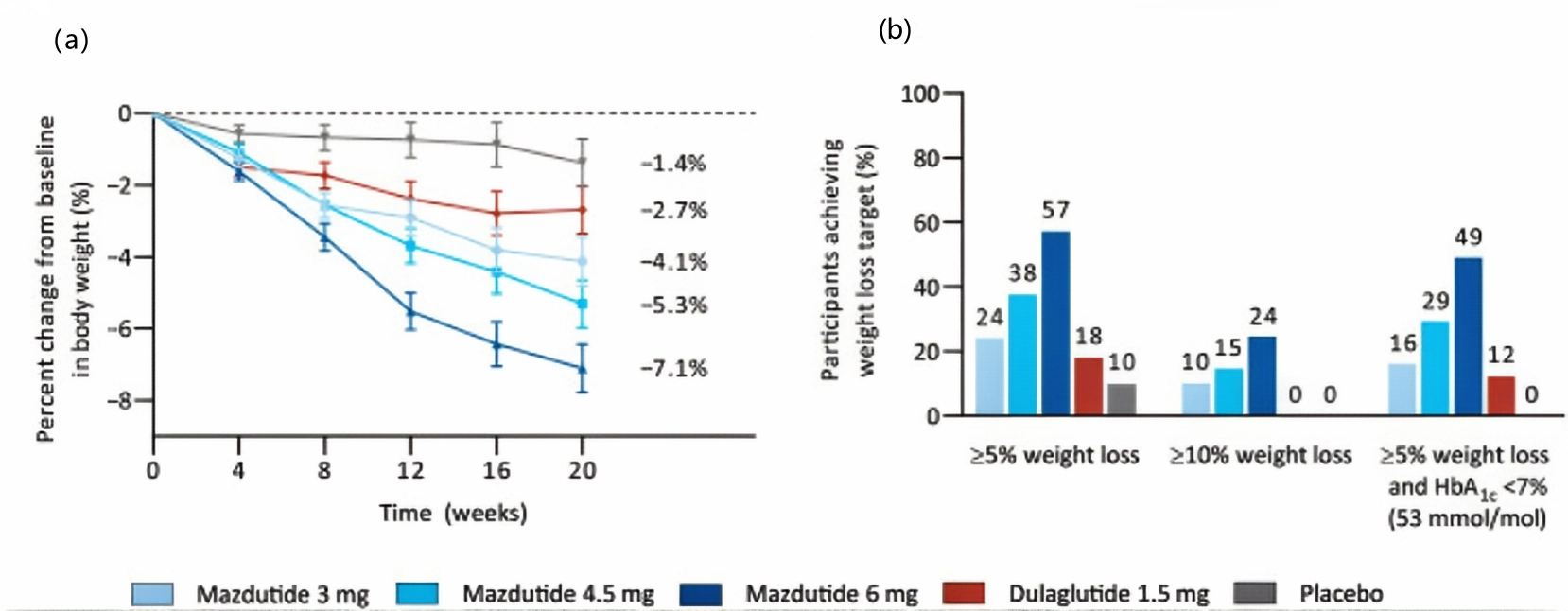
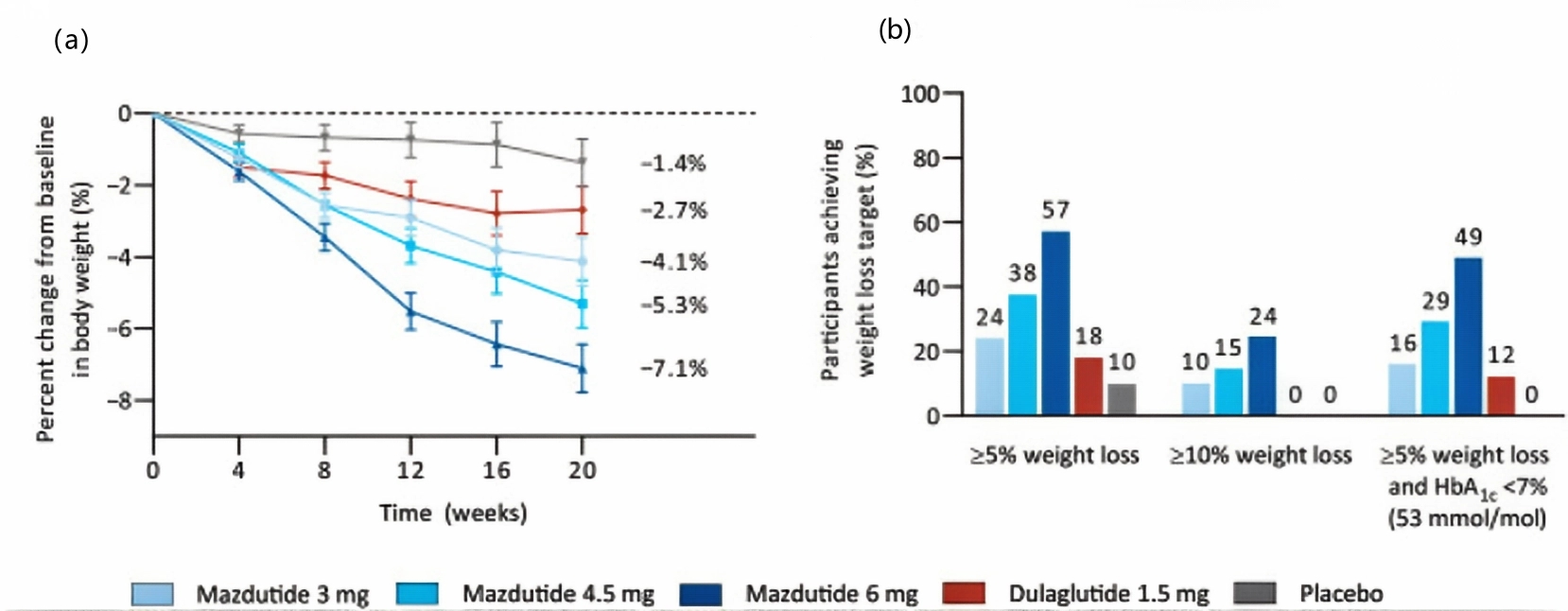
Figure 3 a: Percent change from baseline in body weight over time. b: Proportion of participants reaching weight loss targets.
Application Research
(1) Clinical Trial Progress
Early Clinical Trials: Early Phase 1 clinical trials primarily assessed the safety, tolerability, and preliminary efficacy of Mazdutide. In a randomized, placebo-controlled, multiple-dose escalation Phase 1b trial, adults who were overweight or obese were administered different doses of Mazdutide (up to 10 mg). The results showed that Mazdutide was well tolerated within this dose range, with no serious adverse events reported, and significant weight loss was observed.
Phase 2 clinical trial: The Phase 2 clinical trial further validated the efficacy and safety of Mazdutide in different populations. In a Phase 2 trial targeting Chinese patients with type 2 diabetes, Mazdutide demonstrated significant effects in reducing HbA1c and body weight, with safety comparable to the placebo group. Common adverse reactions were primarily mild to moderate gastrointestinal reactions, such as diarrhea and nausea. In a Phase 2 trial targeting overweight or obese adults, 24 weeks of Mazdutide treatment (up to 6 mg) demonstrated good weight loss efficacy and safety.
(2) Comparative Studies with Other Drugs
Comparison with GLP-1 receptor agonists: Compared to traditional GLP-1 receptor agonists, Mazdutide may have more significant effects on weight loss and blood glucose regulation due to its dual activation of GLP-1 receptors and glucagon receptors. In some studies, Mazdutide achieved greater weight loss than certain GLP-1 receptor agonists within the same treatment period and demonstrated superior stability in blood glucose control.
Comparison with other antidiabetic drugs: Compared with traditional oral antidiabetic drugs, Mazdutide not only effectively lowers blood glucose but also offers additional benefits such as weight loss and improved cardiovascular metabolic markers.
Conclusion
As a GLP-1/glucagon dual receptor agonist, Mazdutide achieves comprehensive regulation of energy metabolism, blood glucose, blood pressure, and blood lipids by simultaneously activating two important metabolic regulatory receptors, demonstrating more comprehensive and powerful therapeutic effects than single receptor agonists. Mazdutide holds significant promise for various metabolic disorders, including obesity, type 2 diabetes, and hyperuricemia.
Sources
[1] Zhang B, Cheng Z, Chen J, et al. Efficacy and Safety of Mazdutide in Chinese Patients With Type 2 Diabetes: A Randomized, Double-Blind, Placebo-Controlled Phase 2 Trial[J]. Diabetes Care, 2024,47(1):160-168.DOI:10.2337/dc23-1287.
[2] Nalisa D L, Cuboia N, Dyab E, et al. Efficacy and safety of Mazdutide on weight loss among diabetic and non-diabetic patients: a systematic review and meta-analysis of randomized controlled trials[J]. Frontiers in Endocrinology, 2024,15. https://api.semanticscholar.org/CorpusID:267984513.
[3] Ji L, Jiang H, Cheng Z, et al. A phase 2 randomised controlled trial of mazdutide in Chinese overweight adults or adults with obesity[J]. Nature Communications, 2023,14(1):8289.DOI:10.1038/s41467-023-44067-4.
[4] Jiang H, Zhang Y, REN Y S. 77-LB: A Novel Glucagon-Like Peptide-1 (GLP-1R) and Glucagon (GCGR) Receptor Dual Agonist, Mazdutide (IBI362), Attenuates Hyperuricemia in Hyperuricemic Rats[J]. Diabetes, 2023. https://api.semanticscholar.org/CorpusID:259452040
[5] Ji L, Gao L, Jiang H, et al. Safety and efficacy of a GLP-1 and glucagon receptor dual agonist mazdutide (IBI362) 9 mg and 10 mg in Chinese adults with overweight or obesity: A randomised, placebo-controlled, multiple-ascending-dose phase 1b trial[J]. Eclinicalmedicine, 2022,54:101691.DOI:10.1016/j.eclinm.2022.101691.
Product available for research use only:



