What are you looking for?
Search
Survodutide's Dual Receptor: Obesity Treatment
ALL ARTICLES AND PRODUCT INFORMATION PROVIDED ON THIS WEBSITE ARE SOLELY FOR INFORMATION DISSEMINATION AND EDUCATIONAL PURPOSES.
The products provided on this website are intended exclusively for in vitro research. In vitro research (Latin: *in glass*, meaning in glassware) is conducted outside the human body. These products are not pharmaceuticals, have not been approved by the U.S. Food and Drug Administration (FDA), and must not be used to prevent, treat, or cure any medical condition, disease, or ailment. It is strictly prohibited by law to introduce these products into the human or animal body in any form.
1. Introduction
Obesity has become a growing public health concern worldwide, significantly increasing the risk of various complications such as cardiovascular disease and diabetes. Finding effective obesity treatment drugs is of utmost importance. Survodutide, as a dual agonist acting on the glucagon receptor (GCGR) and the glucagon-like peptide-1 receptor (GLP-1R), shows great potential in the field of obesity treatment.

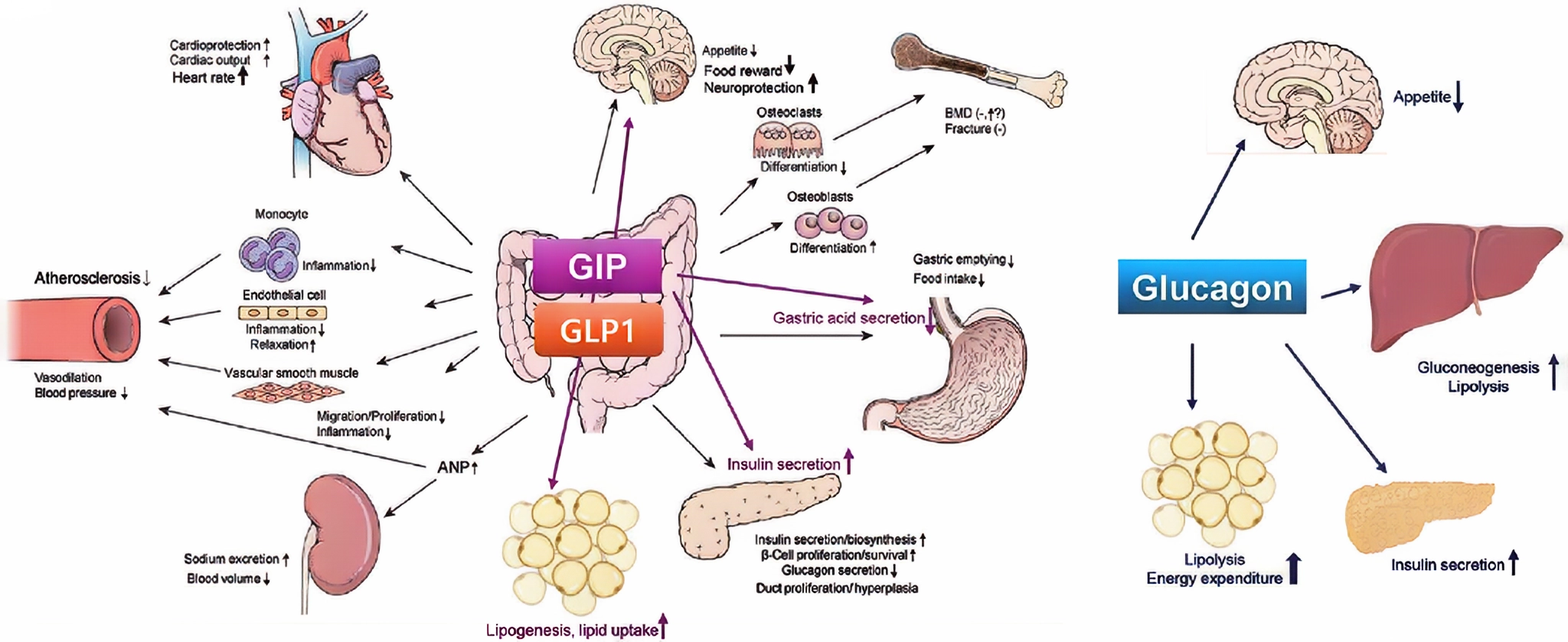
Figure 1 Effects of glucagon-like peptide-1 (GLP-1), glucose-dependent insulinotropic polypeptide (GIP), and glucagon on various vital organs and tissues.
2. Overview of Survodutide
2.1 Development Background
The global incidence of obesity and type 2 diabetes mellitus (T2DM) continues to rise, and traditional antidiabetic drugs often lead to weight gain, prompting an urgent need for novel therapies that can simultaneously treat T2DM and obesity. GLP-1 receptor agonists (GLP-1 RAs) and dual GLP-1/GIP receptor agonists have demonstrated efficacy in weight loss and metabolic improvement, Survodutide (BI 456906) has emerged as a promising candidate, a long-acting dual agonist currently under investigation.
2.2 Target Mechanism
Survodutide's unique feature lies in its ability to simultaneously activate GCGR and GLP-1R. GCGR primarily regulates blood glucose levels, promoting glycogenolysis and gluconeogenesis in the liver upon activation, thereby elevating blood glucose levels. In the mechanism of action of Survodutide, it does not simply raise blood glucose levels but instead regulates energy metabolism through synergistic action with GLP-1R. GLP-1R is widely distributed in multiple tissues and organs, including the pancreas, gastrointestinal tract, heart, and brain. Activating GLP-1R promotes insulin secretion, inhibits glucagon secretion, delays gastric emptying, increases satiety, thereby reducing food intake and lowering body weight. Survodutide exerts a more comprehensive metabolic regulatory effect through dual receptor agonism, aiding in the treatment of obesity.
3. The Role of Survodutide in Obesity Treatment
3.1 Weight Regulation Effect
Multiple clinical studies have demonstrated that Survodutide has a significant weight-reducing effect in obese patients. In a Phase 2 clinical trial targeting obese individuals without diabetes, 387 participants aged 18 to 75 years with a body mass index (BMI) ≥27 kg/m² were randomly assigned to five groups, receiving weekly subcutaneous injections of Survodutide (0.6, 2.4, 3.6, or 4.8 mg) or placebo for 46 weeks. At the end of 46 weeks, all Survodutide dose groups showed weight loss, with the 4.8 mg group achieving an average weight reduction of up to 18.7%. From the subgroup analysis by gender and BMI, women showed a more pronounced weight loss after receiving 4.8 mg of Survodutide, with an average weight loss percentage of -17.0%, while men had -11.9%. Among different BMI subgroups, participants with BMI < 30 kg/m² who received Survodutide 4.8 mg treatment experienced the highest weight loss percentage, reaching -19.1%. This indicates that Survodutide has a weight-reducing effect on obese patients of different genders and BMI levels, with more pronounced effects in certain subgroups.

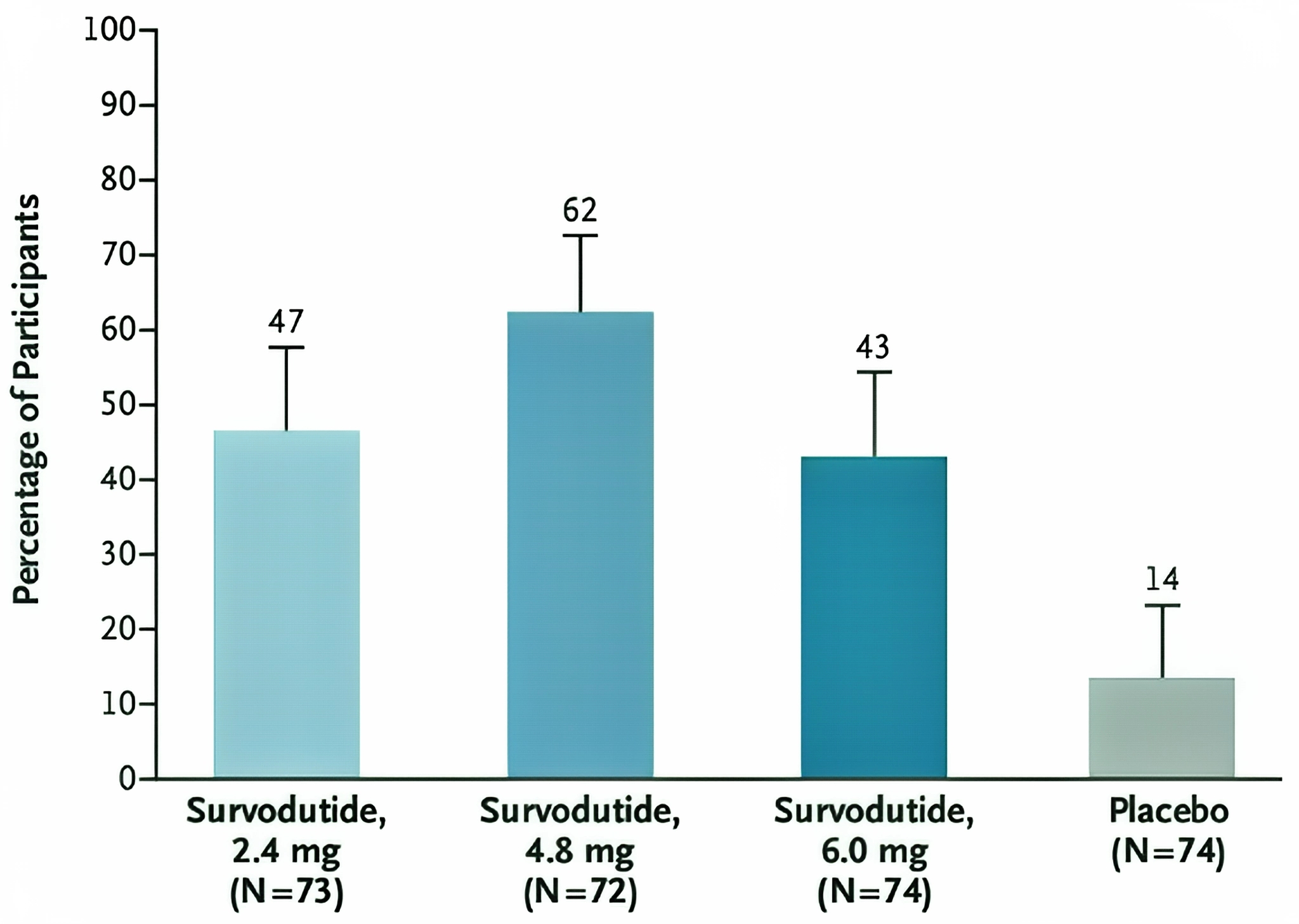
Figure 2 Primary End Point after 48 Weeks of Planned Treatment.
3.2 Effects on Body Fat Distribution
In addition to total weight loss, Survodutide also had beneficial effects on body fat distribution. Compared with the placebo group, all Survodutide dose groups showed a reduction in waist circumference, with the 4.8 mg group achieving the greatest average reduction of 16.6 cm. A reduction in waist circumference indicates a decrease in abdominal fat, which is closely associated with the risk of cardiovascular disease. This suggests that Survodutide not only reduces body weight but also improves body fat distribution by reducing abdominal fat, thereby lowering the risk of obesity-related cardiovascular diseases.
3.3 Improvement in metabolic parameters
Survodutide not only regulates body weight but also has positive effects on multiple metabolic parameters. In terms of lipid metabolism, during the treatment period, triglycerides (TG) decreased significantly in all Survodutide groups, and very low-density lipoprotein (VLDL) decreased slightly in all Survodutide dose groups. LDL decreased in the 2.4 and 3.6 mg dose groups, while total cholesterol (TC) and non-HDL cholesterol (non-HDL-C) decreased in the 0.6, 2.4, and 3.6 mg groups. HDL remained relatively stable throughout treatment. Regarding blood pressure, in actual treatment, Survodutide can reduce systolic blood pressure (SBP) by up to 10.2 mmHg and diastolic blood pressure (DBP) by 4.8 mmHg, with similar blood pressure-lowering effects observed regardless of whether hypertension was present prior to screening. These improvements in metabolic parameters help reduce the risk of cardiovascular disease in obese patients, further highlighting Survodutide's comprehensive advantages in the treatment of obesity.
3.4 Mechanism of Action
Survodutide exerts its weight-loss effects by activating GCGR and GLP-1R. Activation of GLP-1R promotes insulin secretion, enhances insulin sensitivity, suppresses appetite, and reduces food intake. It also delays gastric emptying, prolonging the time food remains in the stomach and further increasing satiety. Activation of GCGR participates in hepatic metabolic regulation by promoting fat oxidation and inhibiting fat synthesis, thereby reducing fat accumulation in the body. Survodutide may also influence energy metabolism and body weight through indirect mechanisms such as regulating the gut microbiota.
4. Application of Survodutide in the Treatment of Obesity
4.1 Clinical Research Evidence
In a study of patients with type 2 diabetes and obesity, Survodutide also demonstrated good hypoglycemic and weight-loss effects. After 16 weeks of treatment, patients' hemoglobin A1c (HbA1c) levels significantly decreased, with a maximum reduction of 1.7%, and body weight also decreased significantly, with a maximum reduction of 14.9%. In a study of patients with metabolic dysfunction-associated steatohepatitis (MASH) and obesity, Survodutide not only improved liver histological markers but also led to weight loss in patients. These studies further confirm the efficacy of Survodutide in various populations with obesity-related conditions.
4.2 Comparative Advantages Over Other Drugs
Compared to traditional weight-loss drugs, Survodutide offers multiple advantages. Traditional weight-loss drugs often target a single mechanism, resulting in limited efficacy and a higher risk of adverse effects. Survodutide, however, acts through dual receptor agonism, effectively reducing body weight while simultaneously improving metabolic parameters such as blood glucose, lipid levels, and blood pressure, thereby providing comprehensive prevention and treatment for obesity-related complications.
Compared to other novel weight-loss drugs such as semaglutide (a GLP-1 receptor agonist), in animal experiments, both Survodutide and semaglutide induced weight loss. However, Survodutide exhibited more unique effects on food preferences and lipid abnormalities. Semaglutid reduces food intake in the early stages of treatment but returns to baseline levels later, while Survodutide continuously reduces intake of high-fat diets and high-fructose water over a 5-week treatment period. Regarding lipid profiles, Semaglutid primarily reduces total cholesterol by lowering HDL-cholesterol, while Survodutide demonstrates cholesterol reduction across all lipoprotein components, including LDL-cholesterol. This suggests that Survodutide may offer superior effects in regulating food intake and lipid profiles.
5. Conclusion
Survodutide, as a novel dual agonist of GCGR/GLP-1R, demonstrates significant efficacy in the treatment of obesity. It not only effectively reduces body weight and improves body fat distribution but also comprehensively regulates multiple metabolic parameters such as blood glucose, blood lipids, and blood pressure, offering multifaceted benefits for patients with obesity.
Sources
[1] Yousif A, Hassan E, Mudarres M F, et al. Survodutide, a new horizon in the treatment of obesity and Type 2 diabetes mellitus: A narrative review[J]. Yemen Journal of Medicine, 2024,3:97-101.DOI:10.18231/j.yjom.2024.005.
[2] Le Roux C W, Steen O, Lucas K J, et al. 6926 Subgroup Analysis by Gender and Body Mass Index (BMI) in People Living With Overweight/Obesity in the Survodutide, a Glucagon/GLP-1 Receptor Dual Agonist, Phase II Trial[J]. Journal of the Endocrine Society, 2024,8(Supplement_1):bvae133-bvae163.DOI:10.1210/jendso/bvae163.033.
[3] Sanyal A J, Bedossa P, Fraessdorf M, et al. A Phase 2 Randomized Trial of Survodutide in MASH and Fibrosis.[J]. The New England Journal of Medicine, 2024.
[4] Briand F, Augustin R, Bleymehl K, et al. 7279 Survodutide and Semaglutid Both Induce Weight Loss but Show Different Effects on Food Preference and Dyslipidemia in the Free Choice Diet-induced Obese Hamster Model[J]. Journal of the Endocrine Society, 2024,8(Supplement_1):bvae134-bvae163.DOI:10.1210/jendso/bvae163.034.
[5] Her M B U, Rosenstock J, Hoefler J, et al. Dose–response effects on HbA1c and bodyweight reduction of survodutide, a dual glucagon/GLP-1 receptor agonist, compared with placebo and open-label semaglutide in people with type 2 diabetes: a randomised clinical trial[J]. Diabetologia, 2023,67:470-482.
Product available for research use only:



