What are you looking for?
Search
SS-31: Repairing Cellular Damage
ALL ARTICLES AND PRODUCT INFORMATION PROVIDED ON THIS WEBSITE ARE SOLELY FOR INFORMATION DISSEMINATION AND EDUCATIONAL PURPOSES.
The products provided on this website are intended exclusively for in vitro research. In vitro research (Latin: *in glass*, meaning in glassware) is conducted outside the human body. These products are not pharmaceuticals, have not been approved by the U.S. Food and Drug Administration (FDA), and must not be used to prevent, treat, or cure any medical condition, disease, or ailment. It is strictly prohibited by law to introduce these products into the human or animal body in any form.
Overview
SS-31, also known as Elamipretide, is a novel aromatic cationic peptide. SS-31 can freely cross the blood-brain barrier, a property that makes it suitable for the treatment of neurological disorders. The blood-brain barrier serves as a crucial protective barrier for the central nervous system, with most drugs struggling to cross it. However, SS-31 overcomes this limitation, directly reaching the site of neural damage and offering hope for the repair of neural cell damage. It belongs to the class of small-molecule peptides, with a relatively low molecular weight, which confers it with good mobility and bioavailability within the body. The cations carried by SS-31 enable it to interact with negatively charged components of biological membranes, particularly the negatively charged inner membrane phospholipids of mitochondria. This specific binding allows SS-31 to accumulate in mitochondria, thereby exerting its regulatory effects on mitochondrial function.

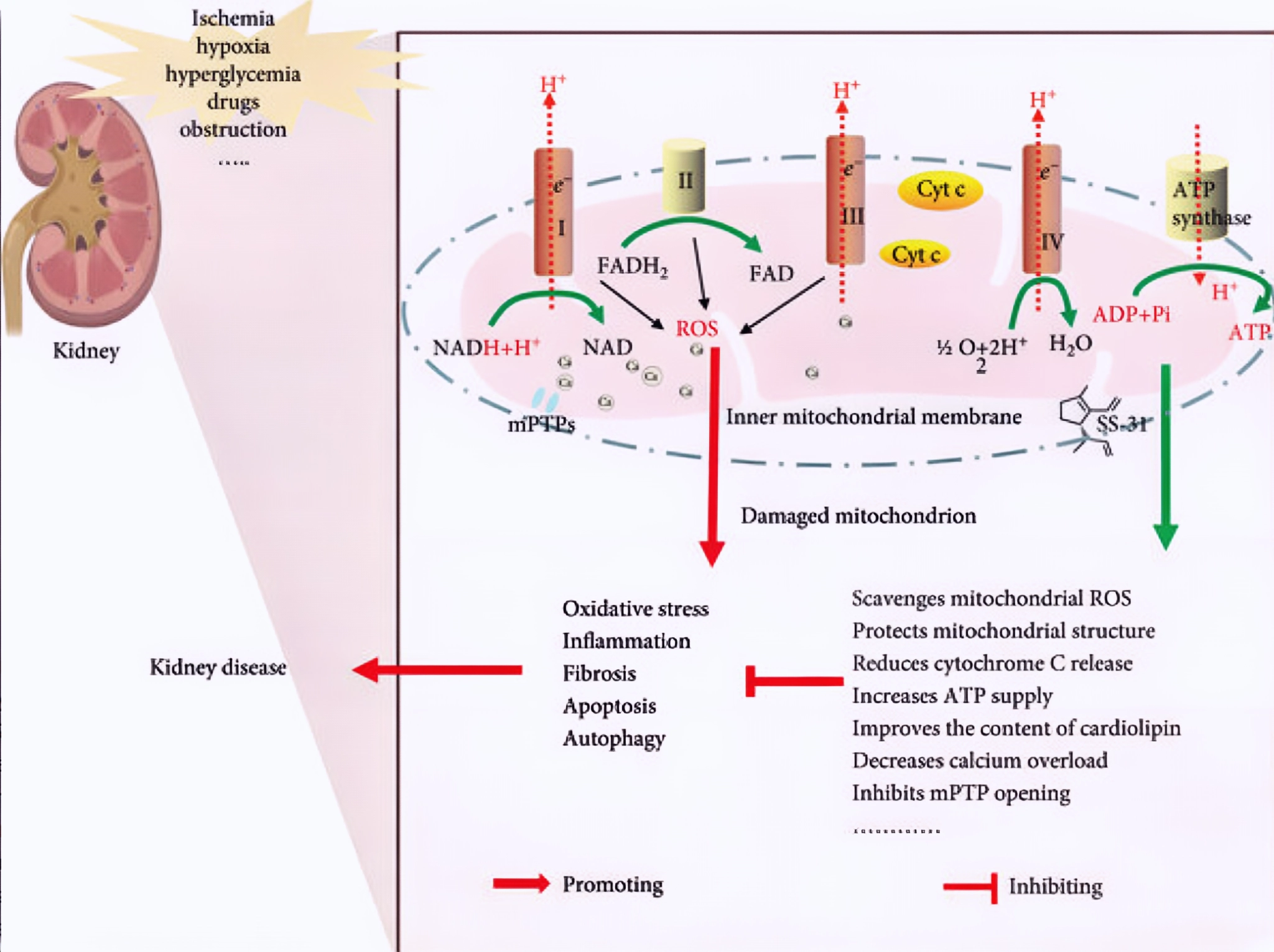
Figure 1 Mechanism of action of SS-31 against kidney disease.
Mechanism of Action
Regulation of Mitochondrial Function
Mitochondria, as the cell's energy powerhouse, play a critical role in cellular survival and metabolism. SS-31, with its cationic structure, can widely accumulate in the negatively charged inner membrane phospholipids of mitochondria, thereby enhancing mitochondrial respiration. The mitochondrial respiratory chain is a critical pathway for cellular energy production (ATP). SS-31 optimizes the function of related proteins in the respiratory chain, improving electron transport efficiency, thereby promoting ATP synthesis and providing cells with sufficient energy to maintain normal physiological activities such as cell division, differentiation, and material transport.
SS-31 activates mitochondrial biogenesis in neurons, promoting the generation of new mitochondria. When cells face damage or stress, the generation of new mitochondria helps replenish damaged or dysfunctional mitochondria, maintaining the health of the mitochondrial population within the cell. It also enhances mitochondrial autophagy, the cellular mechanism for clearing damaged mitochondria. If damaged mitochondria are not promptly cleared, they release large amounts of reactive oxygen species (ROS), further damaging cells. SS-31 effectively reduces ROS production by enhancing mitochondrial autophagy, thereby protecting cells from oxidative stress damage.
Inhibiting inflammatory responses
In many cellular damage processes, inflammatory responses often accompany the damage. Cytoplasmic phospholipase A2 (cPLA2), as the primary mediator of neuroinflammation, breaks down membrane phospholipids at the sn-2 position of lysosomes under pathological conditions, producing lysophospholipids and ω3 polyunsaturated fatty acids, leading to lysosomal membrane permeabilization (LMP) and creating an inflammatory environment. SS-31 can downregulate the expression level of phosphorylated cytoplasmic phospholipase A2 (p-cPLA2), inhibit the phosphorylation of cPLA2, thereby reducing the production of inflammatory mediators and alleviating the damage caused by inflammatory responses to cells.
In studies on sepsis-induced myocardial injury, SS-31 was found to inhibit the activation of NF-κB and NLRP3, thereby improving myocardial inflammatory responses. NF-κB is a key regulator of inflammatory signaling pathways, while NLRP3 is a crucial component of the inflammasome. Their activation leads to the release of a large number of inflammatory factors, such as IL-6, IL-1β, and TNF-α. By inhibiting the activation of these two key factors, SS-31 reduces the expression levels of inflammatory factors, alleviates inflammatory damage to myocardial cells, and maintains their normal function.
Promoting autophagy and inhibiting pyroptosis
Autophagy is an important self-protective mechanism within cells that clears damaged organelles, misfolded proteins, and other harmful substances. In studies of spinal cord injury, it was found that SS-31 enhances the levels of autophagy-related proteins such as Beclin-1, VPS34, and LC3, reduces the levels of autophagy substrate proteins such as p62, but does not affect the levels of lysosomal biogenesis-related proteins such as ATP6V1B2 and LAMP1. This indicates that SS-31 can influence the formation of autophagosomes after spinal cord injury, such as p62, without affecting the levels of lysosomal biogenesis-related proteins, such as ATP6V1B2 and LAMP1. This suggests that SS-31 can influence the formation of autophagosomes after spinal cord injury, promote the normal progression of the autophagy pathway, assist cells in clearing damaged components, and restore the stability of the intracellular environment.
Pyroptosis is an inflammatory programmed necrosis that leads to excessive cellular damage in various diseases. SS-31 reduces the levels of pyroptosis-related proteins, such as ASC, GSDMD, Caspase-1, NLRP3, NLRP1, IL-1β, and IL-18, thereby inhibiting the occurrence of pyroptosis. There is a complex interplay between autophagy and pyroptosis; enhancing autophagy can inhibit pyroptosis. SS-31 regulates this balance, effectively reducing cell death caused by pyroptosis and promoting cellular repair and survival.
Applications
Neurological Diseases
Spinal cord injury is a severe disabling condition leading to permanent functional loss and motor impairments. Due to the presence of the blood-brain barrier, conventional drugs struggle to reach the injured site. SS-31, with its ability to cross the blood-brain barrier, shows promising potential in the treatment of spinal cord injury. When administered via intraperitoneal injection to mice with spinal cord injury, behavioral assessments using the BMS mouse scale, gait analysis, and inclined plane test revealed that it significantly promotes functional recovery post-injury. Histologically, using HE, Masson, MAP2, and SYN staining, it was determined that SS-31 reduces the area of spinal cord glial scarring, increases the number of dendrites and synapses, and promotes the structural and functional repair of spinal cord neurons.
In studies of some neurodegenerative diseases, such as Parkinson's disease and Alzheimer's disease, SS-31 has also shown potential therapeutic value. These diseases are often accompanied by oxidative stress-induced neuronal damage, inflammatory responses, and mitochondrial dysfunction. SS-31, through multiple mechanisms such as regulating mitochondrial function, inhibiting inflammatory responses, and promoting cellular autophagy, holds promise for delaying neuronal degeneration and improving patient symptoms.
Cardiovascular Diseases
Cardiac dysfunction is a common and life-threatening complication of sepsis. SS-31 exhibits protective effects against myocardial injury in sepsis. In in vivo experiments, mice in the SS-31 intervention group showed significantly improved myocardial tissue disorganization and inflammatory infiltration compared to the sepsis group, and a significant reduction in apoptotic cells. SS-31 can inhibit the upregulation of inflammatory factors in myocardial tissue, increase ATP content, improve redox status, maintain mitochondrial membrane potential, and inhibit the activation of NF-κBp65 and NLRP3, thereby alleviating LPS-induced myocardial injury. This suggests that SS-31 may become a potential drug for treating sepsis-induced cardiomyopathy.
In other cardiovascular diseases, such as myocardial ischemia-reperfusion injury, SS-31 may also play an important role. The myocardial ischemia-reperfusion process leads to the production of a large amount of ROS, triggering inflammatory responses and cell apoptosis.
Conclusion
SS-31 can penetrate the blood-brain barrier, target mitochondria to repair cellular damage, and has potential applications in neurological diseases, cardiovascular diseases, skeletal muscle repair, and diabetic vascular endothelial injury. By targeting mitochondria as the core target, it offers a new therapeutic theory.
Sources
[1] Zhu Y, Luo M, Bai X, et al. SS-31, a Mitochondria-Targeting Peptide, Ameliorates Kidney Disease[J]. Oxidative Medicine and Cellular Longevity, 2022,2022:1295509.DOI:10.1155/2022/1295509.
Product available for research use only:



