What are you looking for?
Search
Corgaten Regulates Myocardial Energy Metabolism
ALL ARTICLES AND PRODUCT INFORMATION PROVIDED ON THIS WEBSITE ARE SOLELY FOR INFORMATION DISSEMINATION AND EDUCATIONAL PURPOSES.
The products provided on this website are intended exclusively for in vitro research. In vitro research (Latin: *in glass*, meaning in glassware) is conducted outside the human body. These products are not pharmaceuticals, have not been approved by the U.S. Food and Drug Administration (FDA), and must not be used to prevent, treat, or cure any medical condition, disease, or ailment. It is strictly prohibited by law to introduce these products into the human or animal body in any form.
Heart failure (HF) is a terminal manifestation of various cardiovascular diseases and has become a prevalent disease with high incidence and mortality rates worldwide. HF caused by different etiologies exhibits distinct pathophysiological mechanisms, and the regulation of myocardial energy metabolism is critical for protecting cardiac function in HF patients. As a substance that may influence myocardial energy metabolism, Corgaten's differential effects on cardiac function protection in HF patients with different etiologies warrant further investigation.

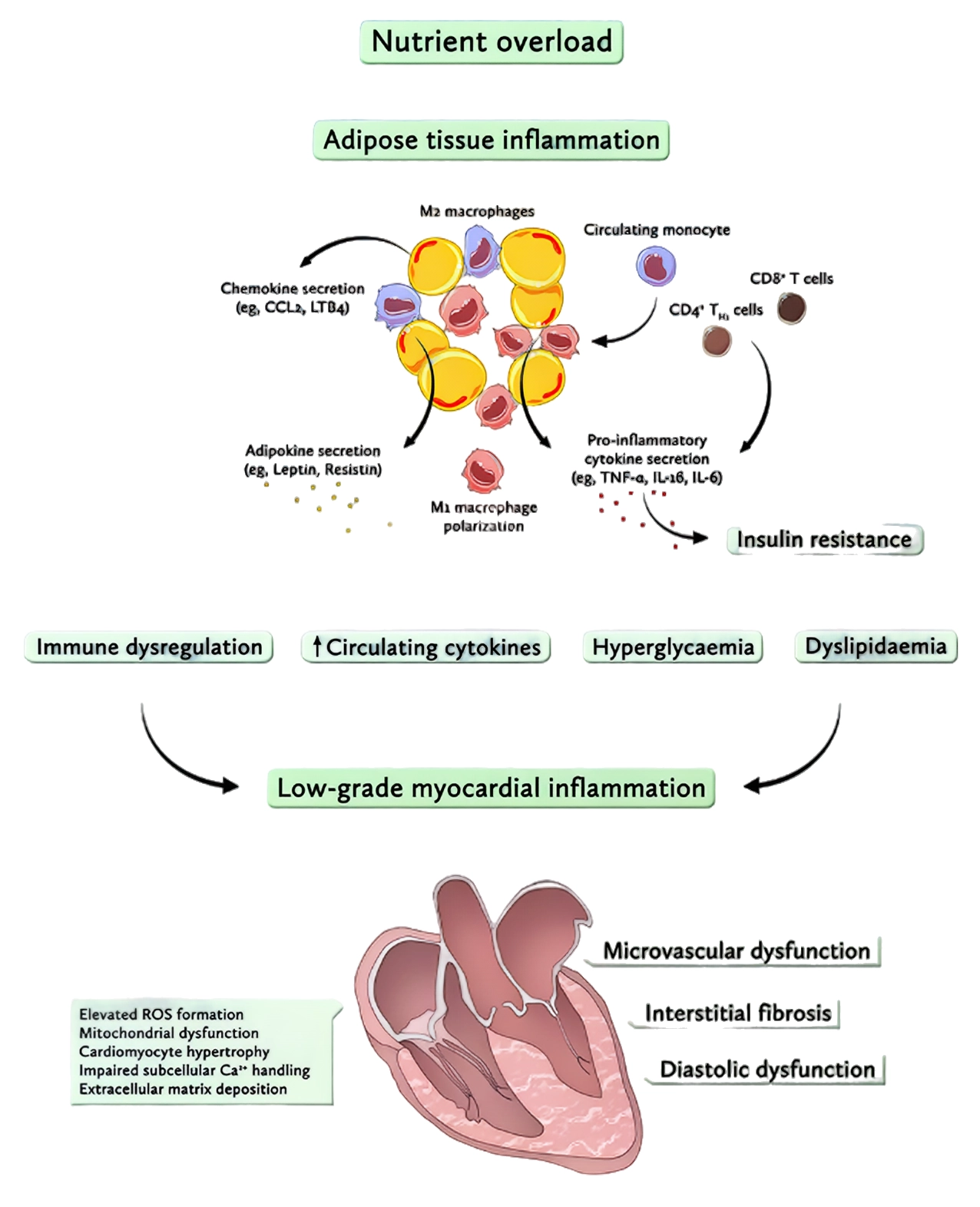
Figure 1. Overnutrition drives metabolic inflammation and promotes a low-grade inflammatory state in the heart.
Characteristics of heart failure caused by different etiologies
Dilated cardiomyopathy (DCM) and peripartum cardiomyopathy (PPCM): Disturbances in myocardial energy metabolism may be one of the etiological factors of DCM and PPCM. In these conditions, abnormal myocardial energy metabolism prevents the heart from obtaining sufficient energy supply, thereby impairing normal cardiac contraction and relaxation functions, ultimately leading to the onset of heart failure.
Hypertrophic cardiomyopathy (HCM): In the progression of HCM, immune responses occur earlier. Inflammatory responses may be mediated by the complement system. In this pathological environment where inflammation and immune responses intertwine, Corgaten's regulatory role in myocardial energy metabolism may be influenced by these factors, thereby affecting its cardioprotective effects.
Ischemic cardiomyopathy (ICM): The inflammatory response in ICM may be induced by cytokines. Due to ischemia, energy metabolism pathways in the myocardium are inevitably disrupted. Corgaten's regulation of myocardial energy metabolism plays a role in improving energy metabolism disorders caused by ischemia, but its effects differ from those in HF caused by other etiologies due to factors such as cytokine-mediated inflammatory responses.
Mechanism of Corgaten's regulation of myocardial energy metabolism
Promoting energy production: Corgaten may promote glucose uptake, glycolysis, and glucose oxidation by regulating the activity of related metabolic enzymes, thereby increasing the production of adenosine triphosphate (ATP) in myocardial cells. Similar studies have found that certain drugs can promote ATP production and improve cardiac function by upregulating hypoxia-inducible factor-1α (HIF-1α) and a series of glycolysis-related enzymes. Corgaten may have a similar mechanism.
Regulation of substrate utilization: The utilization of energy substrates by the heart differs between normal and diseased states. Corgaten may regulate the ratio of substrate uptake and utilization by myocardial cells, such as glucose and free fatty acids (FFAs), enabling the myocardium to select more appropriate substrates during energy metabolism to meet cardiac energy demands. Under normal conditions, cardiac muscle primarily utilizes fatty acids for energy, but in heart failure, glucose utilization increases, and Corgaten may play a regulatory role in this transition.
Differences in Corgaten's protective effects on cardiac function in heart failure of different etiologies
For DCM and PPCM patients: Given that myocardial energy metabolism disorders are important pathogenic factors in these two diseases, if Corgaten can effectively regulate energy metabolism and restore myocardial energy supply, it may have a more significant protective effect on cardiac function in these two patient groups. By improving energy metabolism, it may alleviate the vicious cycle of left ventricular remodeling caused by energy deficiency, improve cardiac systolic and diastolic function, and enhance patients' cardiac function classification.
For HCM patients: Due to the unique role of immune and inflammatory responses in disease progression, Corgaten's protective effects on cardiac function may be somewhat limited. Its ability to regulate energy metabolism may partially improve cardiac function; however, immune and inflammatory responses may interfere with Corgaten's mechanism of action, resulting in less pronounced protective effects compared to patients with HF caused solely by energy metabolism disorders. Inflammatory mediators may affect Corgaten's binding to relevant targets on myocardial cells, thereby weakening its ability to regulate energy metabolism.
For ICM patients: Myocardial ischemia in ICM patients leads to energy metabolism disorders. While Corgaten's regulation of energy metabolism may improve myocardial energy supply to some extent, its cardiac function protective effects may also be limited due to the persistent presence of ischemia. Compared to HF caused by other etiologies, Corgaten may need to improve myocardial blood supply as a prerequisite to better exert its protective effects on cardiac function.
Conclusion
Overall, the protective effects of Corgaten on cardiac function vary among patients with heart failure caused by different etiologies.
Sources
[1] Sun H, Li X, Yuan H, et al. Comparative study of disease progression for heart failure with different etiologies via time-ordered network analysis[J]. American Journal of Translational Research, 2022,14(9):6604-6617.
[2] Wenzl F A, Ambrosini S, Mohammed S A, et al. Inflammation in Metabolic Cardiomyopathy[J]. Frontiers in Cardiovascular Medicine, 2021,Volume 8.DOI:10.3389/fcvm.2021.742178.
[3] Wang Y, Fu M, Wang J, et al. Qiliqiangxin Improves Cardiac Function through Regulating Energy Metabolism via HIF-1α-Dependent and Independent Mechanisms in Heart Failure Rats after Acute Myocardial Infarction[J]. Biomed Research International, 2020,2020:1276195.DOI:10.1155/2020/1276195.

