What are you looking for?
Search
Bam15: Regulating Metabolic Function
ALL ARTICLES AND PRODUCT INFORMATION PROVIDED ON THIS WEBSITE ARE SOLELY FOR INFORMATION DISSEMINATION AND EDUCATIONAL PURPOSES.
The products provided on this website are intended exclusively for in vitro research. In vitro research (Latin: *in glass*, meaning in glassware) is conducted outside the human body. These products are not pharmaceuticals, have not been approved by the U.S. Food and Drug Administration (FDA), and must not be used to prevent, treat, or cure any medical condition, disease, or ailment. It is strictly prohibited by law to introduce these products into the human or animal body in any form.
Overview
Bam15, as a selective mitochondrial uncoupler, has been applied in metabolic-related research fields in recent years, offering new treatment options for various metabolic diseases. Mitochondria play a central role in cellular energy metabolism, responsible for generating ATP through oxidative phosphorylation, thereby providing energy for various cellular activities. In certain pathological states, mitochondrial function may become impaired, leading to metabolic disorders and being closely associated with the onset and progression of diseases such as obesity, diabetes, and non-alcoholic fatty liver disease.
Bam15 specifically targets mitochondria and, through a unique mechanism of action, disrupts the tight coupling between electron transport and ATP synthesis. It promotes the backflow of protons across the inner mitochondrial membrane, thereby dissipating the proton gradient, allowing the mitochondrial respiratory chain to continue functioning but reducing the efficiency of ATP synthesis. This uncoupling effect has important physiological significance, as it promotes mitochondrial respiration, increases energy consumption, and dissipates energy in the form of heat, thereby regulating the body's energy metabolism balance.

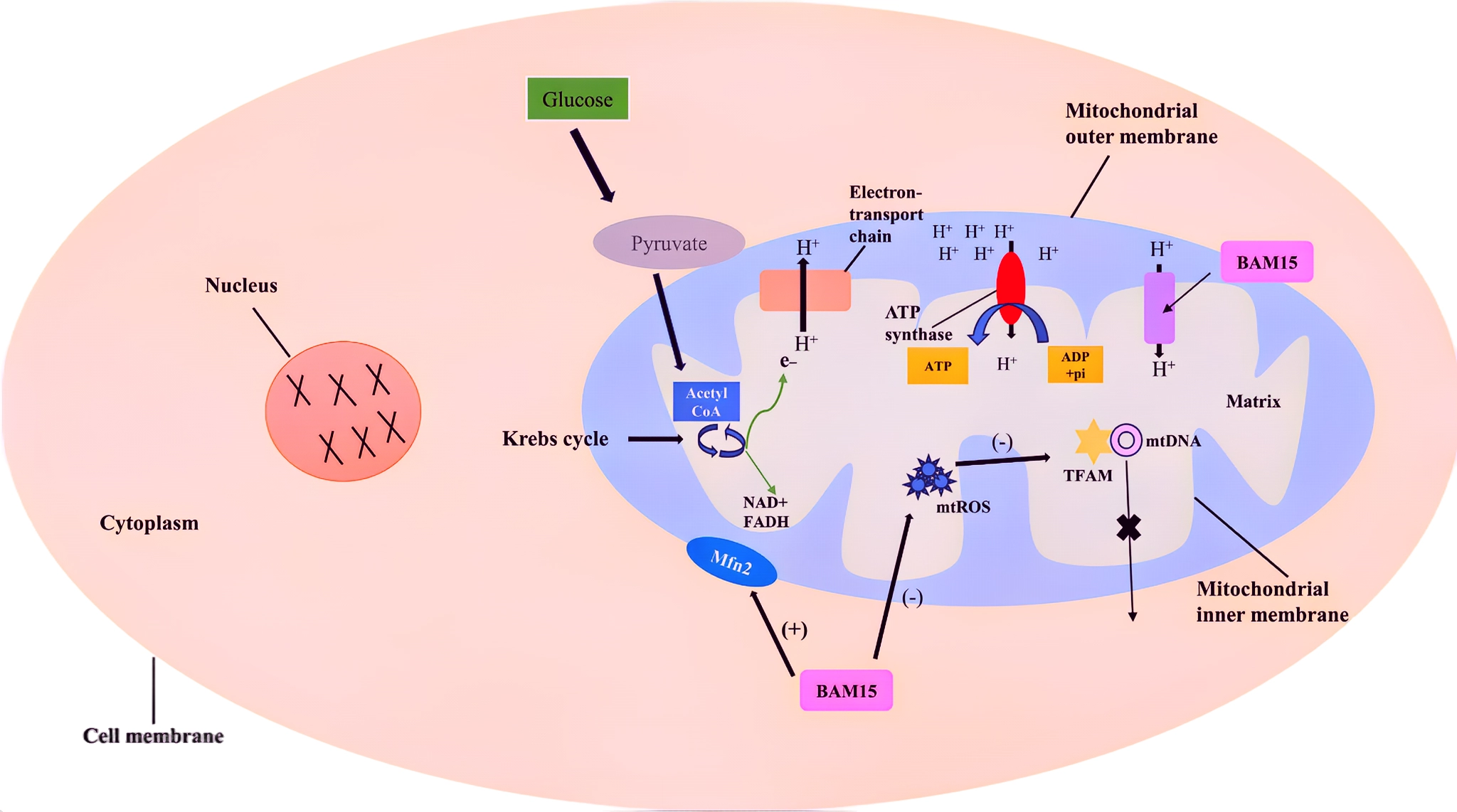
Figure 1 Mechanism of BAM15-induced uncoupling of oxidative phosphorylation.
Effects
Promoting energy metabolism: Bam15 significantly enhances the body's energy metabolism levels through mitochondrial uncoupling mechanisms. Under normal physiological conditions, cellular energy metabolism remains in a relatively stable state to sustain normal physiological functions. When the body ingests excessive energy that cannot be promptly consumed, the surplus energy is stored as fat, leading to weight gain and obesity. Bam15 disrupts this balance by activating mitochondrial respiration, enabling substrates within cells to be oxidized and decomposed more rapidly, thereby increasing energy expenditure and reducing fat accumulation. In animal experiments, mice treated with Bam15 exhibited significantly suppressed weight gain even under a high-fat diet, clearly demonstrating Bam15's role in promoting energy metabolism.
Improving insulin resistance: Insulin resistance is one of the key pathophysiological mechanisms underlying the development of type 2 diabetes. In an insulin-resistant state, the sensitivity of body tissues to insulin decreases, preventing insulin from effectively promoting glucose uptake and utilization, thereby leading to elevated blood glucose levels. Bam15 can improve insulin resistance through multiple pathways. It promotes energy metabolism, reducing excessive fat accumulation in tissues, particularly visceral fat. Excessive fat tissue secretes large amounts of inflammatory factors and adipokines, which interfere with the normal transmission of insulin signaling pathways, leading to insulin resistance. After reducing fat accumulation, Bam15 decreases inflammatory responses, thereby improving the function of the insulin signaling pathway. Bam15 can also directly act on intracellular insulin signaling molecules, enhancing insulin signal transmission and increasing cellular sensitivity to insulin, enabling cells to better uptake and utilize glucose, thereby lowering blood glucose levels.
Promoting mitochondrial autophagy: Mitochondrial autophagy is an important mechanism for cells to maintain mitochondrial quality and function. When mitochondria are damaged or dysfunctional, cells initiate the mitochondrial autophagy process, encapsulating damaged mitochondria and transporting them to lysosomes for degradation to maintain the health of the mitochondrial population within the cell. Under pathological conditions such as a high-fat diet, mitochondria are prone to damage, leading to mitochondrial dysfunction and further exacerbating metabolic disorders. Bam15 significantly promotes mitochondrial autophagy by upregulating the expression of mitochondrial autophagy marker genes Pink1 and Parkin, prompting cells to promptly clear damaged mitochondria, improve mitochondrial quality control, restore normal mitochondrial function, and maintain cellular energy metabolic homeostasis.
Regulation of lipid metabolism: Bam15 plays a crucial role in regulating lipid metabolism. It promotes lipolysis in hepatocytes, inhibits lipid synthesis, and reduces lipid accumulation in the liver. In pathological states such as hyperlipidemia, elevated lipid levels in the blood lead to excessive lipid accumulation in tissues like the liver, triggering lipid-related pathologies such as steatosis. Bam15 modulates the activity of enzymes involved in lipid metabolism, such as promoting the expression of enzymes related to fatty acid oxidation and inhibiting the activity of fatty acid synthesis enzymes, thereby increasing fatty acid oxidation and decomposition while reducing synthesis. This lowers lipid levels in the liver and blood, improving lipid metabolism disorders.
Applications
Treatment of obesity: Obesity has become a global public health issue and is closely associated with the onset of various chronic diseases. Due to its ability to promote energy metabolism and reduce fat accumulation, Bam15 shows great potential in the treatment of obesity. Animal experiment results show that obese mice treated with Bam15 experienced significant weight loss, improved body composition, reduced fat content, and relatively increased lean muscle content. Thus, Bam15 can serve as a potential therapeutic agent for obesity treatment by regulating energy balance, aiding weight loss in obese patients, and reducing the risk of obesity-related complications.
Diabetes Treatment: Given Bam15's significant role in improving insulin resistance and regulating blood glucose levels, it has garnered considerable attention in diabetes therapy. Preclinical studies have shown that Bam15 can enhance insulin sensitivity in diabetic animal models, lower blood glucose levels, and improve impaired glucose tolerance. This provides a new approach and potential drug option for diabetes treatment. Compared to traditional diabetes medications, Bam15 regulates metabolism through a unique mechanism, effectively controlling blood glucose levels without causing severe side effects such as hypoglycemia, thereby offering better treatment outcomes for diabetic patients.
Treatment of non-alcoholic fatty liver disease: Non-alcoholic fatty liver disease is a common metabolic liver disease characterized by excessive lipid accumulation in the liver. Bam15 can effectively reduce hepatic steatosis and improve liver function by regulating lipid metabolism and promoting mitochondrial autophagy. In animal experiments, non-alcoholic fatty liver disease model animals treated with Bam15 showed a significant reduction in liver lipid content and improved liver function markers.
Summary
Bam15 targets mitochondrial metabolism, playing a role in key processes such as energy imbalance, insulin resistance, lipid disorders, and mitochondrial quality control, thereby offering new therapeutic strategies for metabolic diseases such as obesity, diabetes, and fatty liver disease, as well as their associated complications.
Sources
[1] Xiong G, Zhang K, Ma Y, et al. BAM15 as a mitochondrial uncoupler: a promising therapeutic agent for diverse diseases[J]. Frontiers in Endocrinology, 2023,14:1252141.DOI:10.3389/fendo.2023.1252141.
[1] Paris V, Alexopoulos S, HU Y, et al. Mitochondrial Uncoupler BAM15 Ameliorates Associated Metabolic PCOS Traits in a Hyperandrogenic PCOS Mouse Model[J]. Fertility & Reproduction, 2022,04:208.DOI:10.1142/S2661318222741194.
Product available for research use only:



